
PDF Publication Title:
Text from PDF Page: 022
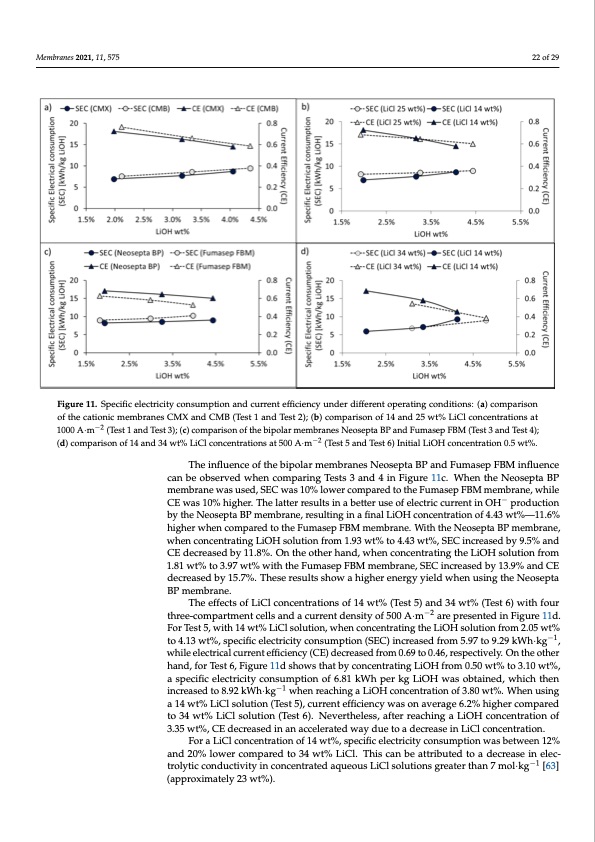
Membranes 2021, 11, 575 22 of 29 Membranes 2021, 11, x FOR PEER REVIEW 23 of 30 Figure 11. Specific electricity consumption and current efficiency under different operating conditions: (a) comparison of Figure 11. Specific electricity consumption and current efficiency under different operating conditions: (a) comparison the cationic membranes CMX and CMB (Test 1 and Test 2); (b) comparison of 14 and 25 wt% LiCl concentrations at 1000 of the cationic membranes CMX and CMB (Test 1 and Test 2); (b) comparison of 14 and 25 wt% LiCl concentrations at A∙m−2 (Test 1 and Test 3); (c) comparison of the bipolar membranes Neosepta BP and Fumasep FBM (Test 3 and Test 4); 1000 A·m−2 (Test 1 and Test 3); (c) comparison of the bipolar membranes Neosepta BP and Fumasep FBM (Test 3 and Test 4); (d) comparison of 14 and 34 wt% LiCl concentrations at 500 A∙m−2 (Test 5 and Test 6) Initial LiOH concentration 0.5 wt%. (d) comparison of 14 and 34 wt% LiCl concentrations at 500 A·m−2 (Test 5 and Test 6) Initial LiOH concentration 0.5 wt%. According to our results, process energy efficiency is affected with increasing LiOH The influence of the bipolar membranes Neosepta BP and Fumasep FBM influence concentration according to OH− leakage effects and cation membrane deswelling. On the can be observed when comparing Tests 3 and 4 in Figure 11c. When the Neosepta BP other hand, increasing HCl solution concentration promotes a higher leakage of Cl− ions membrane was used, SEC was 10% lower compared to the Fumasep FBM membrane, while into the LiOH compartment, affecting current efficiency in the bipolar mem−brane in the CE was 10% higher. The latter results in a better use of electric current in OH production generation of OH− ions, which also affects solution purity. by the Neosepta BP membrane, resulting in a final LiOH concentration of 4.43 wt%—11.6% Table 8 presents a summary of results obtained in long-running tests. The best result higher when compared to the Fumasep FBM membrane. With the Neosepta BP membrane, was achieved in Test 2, where a Neosepta BP membrane, CMB cationic membrane and when concentrating LiOH solution from 1.93 wt% to 4.43 wt%, SEC increased by 9.5% and current density of 1000 A∙m−2 were used. The possibility was shown of achieving LiOH CE decreased by 11.8%. On the other hand, when concentrating the LiOH solution from solutions between 3.34 and 4.35 wt% concentration, with a range of 96.0–95.4% purity. 1.81 wt% to 3.97 wt% with the Fumasep FBM membrane, SEC increased by 13.9% and CE Product purity is therefore inversely proportional to the final concentration obtained. The decreased by 15.7%. These results show a higher energy yield when using the Neosepta lowest average specific electricity consumption (SEC) was achieved in Test 1 between 6.94 BP membrane. and 8.71 kWh per kilogram of LiOH. On the other hand, the highest electrical current The effects of LiCl concentrations of 14 wt% (Test 5) and 34 wt% (Test 6) with four efficiency (CE) was achieved in Test 2, in the range of 0.77–0.59, with an SEC between 7.57 three-compartment cells and a current density of 500 A·m−2 are presented in Figure 11d. and 9.45 kWh per kilogram of LiOH. For Test 5, with 14 wt% LiCl solution, when concentrating the LiOH solution from 2.05 wt% −1 The CMB membrane presented a high current efficiency despite its higher electrical to 4.13 wt%, specific electricity consumption (SEC) increased from 5.97 to 9.29 kWh·kg , resistance, which can be attributed to a higher resistance to co-ion leakage. However, it is while electrical current efficiency (CE) decreased from 0.69 to 0.46, respectively. On the other nhoatnrde,cfomrTmeesnt6d,eFdigtourues1e1idtwshiothwLsitChlatsoblyutcioncsehnitgrahteinrgthLainO1H4fwrotm%.0T.5h0iswmt%emtobr3a.1n0ewta%s, taesstpedecinficloenlegc-rtruincintyincgotnesutsmaptt2i5onwot%f6L.8iC1lkcWonhcepnetrraktgioLnisO,sHhowaisngobdtaminaegde,twohiticshsttrhuecn- −1 tiunrcereafstedr two o8.9h2oukWrsho·fkpgrocweshseingredaucehitnogthaeLhiOigHhcosnmceontitcraptiroenssoufr3e.8re0awcht%ed.,Wcahuesninugsiintgs rau1p4tuwret%anLdiCcolnsotalmutinoanti(oTnesbte5t)w,ceuenrrLeniCtleaffincdieLniOcyHwsaosluotnioanvse.rCaognes6e.q2u%enhtilgyh,ethrecsoemrepsaurletds atroe3n4owt pt%resLeinCtelds.olution (Test 6). Nevertheless, after reaching a LiOH concentration of 3.35 wt%, CE decreased in an accelerated way due to a decrease in LiCl concentration. For a LiCl concentration of 14 wt%, specific electricity consumption was between 12% and 20% lower compared to 34 wt% LiCl. This can be attributed to a decrease in elec- trolytic conductivity in concentrated aqueous LiCl solutions greater than 7 mol·kg−1 [63] (approximately 23 wt%).PDF Image | Bipolar Membrane Electrodialysis for LiOH Production
PDF Search Title:
Bipolar Membrane Electrodialysis for LiOH ProductionOriginal File Name Searched:
membranes-11-00575-v2.pdfDIY PDF Search: Google It | Yahoo | Bing
Product and Development Focus for Infinity Turbine
ORC Waste Heat Turbine and ORC System Build Plans: All turbine plans are $10,000 each. This allows you to build a system and then consider licensing for production after you have completed and tested a unit.Redox Flow Battery Technology: With the advent of the new USA tax credits for producing and selling batteries ($35/kW) we are focussing on a simple flow battery using shipping containers as the modular electrolyte storage units with tax credits up to $140,000 per system. Our main focus is on the salt battery. This battery can be used for both thermal and electrical storage applications. We call it the Cogeneration Battery or Cogen Battery. One project is converting salt (brine) based water conditioners to simultaneously produce power. In addition, there are many opportunities to extract Lithium from brine (salt lakes, groundwater, and producer water).Salt water or brine are huge sources for lithium. Most of the worlds lithium is acquired from a brine source. It's even in seawater in a low concentration. Brine is also a byproduct of huge powerplants, which can now use that as an electrolyte and a huge flow battery (which allows storage at the source).We welcome any business and equipment inquiries, as well as licensing our turbines for manufacturing.| CONTACT TEL: 608-238-6001 Email: greg@infinityturbine.com | RSS | AMP |